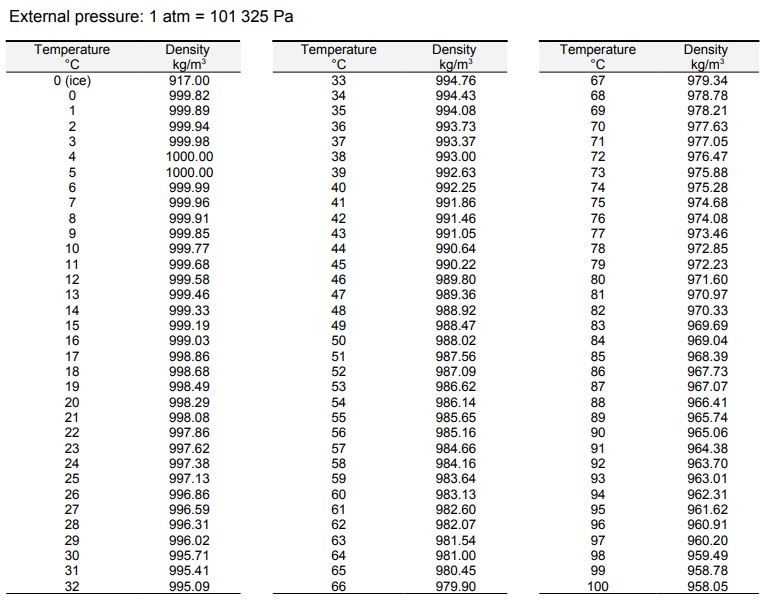
F4) Whole degrees are listed down the left hand side of the table, while tenths of a degree are listed across. Just above and below 4☌ they are almost the same. Temperature (☌) (from Handbook of Chemistry and Physics, 53rd Edition, p. Water samples just above and below 0☌ are very different. At that T, the tendency toward settling into a special low-energy structure runs away in a cooperative manner, and ice crystals form. 4☌ just happens to be the T at which the expansion, due to settling into the special low-energy states, and the contraction, due to the general trend to spend more time in contact at low T, cancel.Ġ☌ is another matter. Nothing really special happens there in the structure of the water. At higher T, the molecules sample even higher energy states, and a majority of them are loosely packed, so the liquid expands- the more typical behavior. When things cool, they settle into lower energy arrangements. For most molecules, that typically involves squeezing together a bit, but water happens to have a low-energy arrangement that’s rather open. The expansion of water at lower T results from the water molecules arranging themselves to minimize the energy of their interactions. I assume you are referring to liquid water, not ice, since 4☌ is about the temperature (T) at which liquid water has a minimum volume, at atmospheric pressure. ĭensity of crude oil, Density of fuel oils, Density of lubricating oil and Density of jet fuel as function of temperature.Īs shown in the figures, the change in density is not linear with temperature - this means that the volumetric expansion coefficient for water is not constant over the temperature range.Rohan- That’s a very nice question. is the density of water at a reference temperature (often taken as 4 degrees Celsius, which is. To calculate the density of water at different temperatures, you can use the following formula: 1 (T T) Where: is the density of water at the desired temperature. The density in units oz/in 3 can with the converting value from above be calculated to 998.21 kg/m 3 0.0005780 (oz/in 3 )/(kg/m 3 ) 0. The density of water changes with temperature. įor other substances, see density and specific weight of acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane and toluene. Water density at temperature 20 o C is 998.21 kg/m 3 (table above). See also other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium. See Water and Heavy Water for thermodynamic properties at standard condtions. Most animals and plants contain more than 60 water by volume. base temperature as 60F.) Water is essential for life. At 4C pure water has a specific gravity of 1. The density of water depends on temperature and pressure as shown below: At 4C pure water has a density (weight or mass) of about 1 g/cu.cm, 1 g/ml, 1 kg/litre, 1000 kg/cu.m, 1 tonne/cu.m or 62.4 lb/cu.ft. 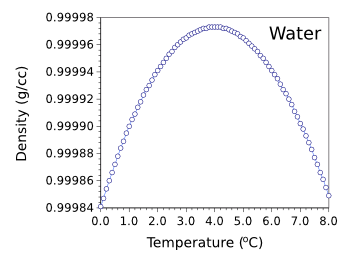
Note! Temperature must be within the ranges 0-370 ☌, 32-700 ☏, 273-645 K and 492-1160 °R to get valid values. The output density is given as g/cm 3, kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to calculate the liquid water density at given temperatures. See more about the difference between mass and weight Online Water density Calculator The density of water is 1.940 sl/ft 3at 39 ☏ (4 ☌), and the specific weight in Imperial units is 
The density of water (H 2 O) changes depending on temperature and pressure - as with other substances. In the Imperial system the mass unit is the slug, and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it :ġ = 1 * 1 and 1 = 1 /1 Data chart with the density of water at different temperatures. In the SI system, specific weight of water at 4☌ will be: G = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 m/s 2or 32.17405 ft/s 2 Specific weight is the ratio of the weight to the volume of a substance: Pure water has its highest density 1000 kg/m 3 or 1.940 slug/ft 3 at temperature 4☌ (=39.2☏). Density is the ratio of the mass to the volume of a substance:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
